CAR-T for T Cell Leukaemia
T-ALL is an aggressive blood cancer with poor outcomes after relapse. CAR-T therapy for T-ALL is uniquely challenging: CAR-T cells and leukaemic T cells share surface antigens, causing fratricide and making it difficult to distinguish the therapy from the disease.
We have developed multiple strategies to overcome this:
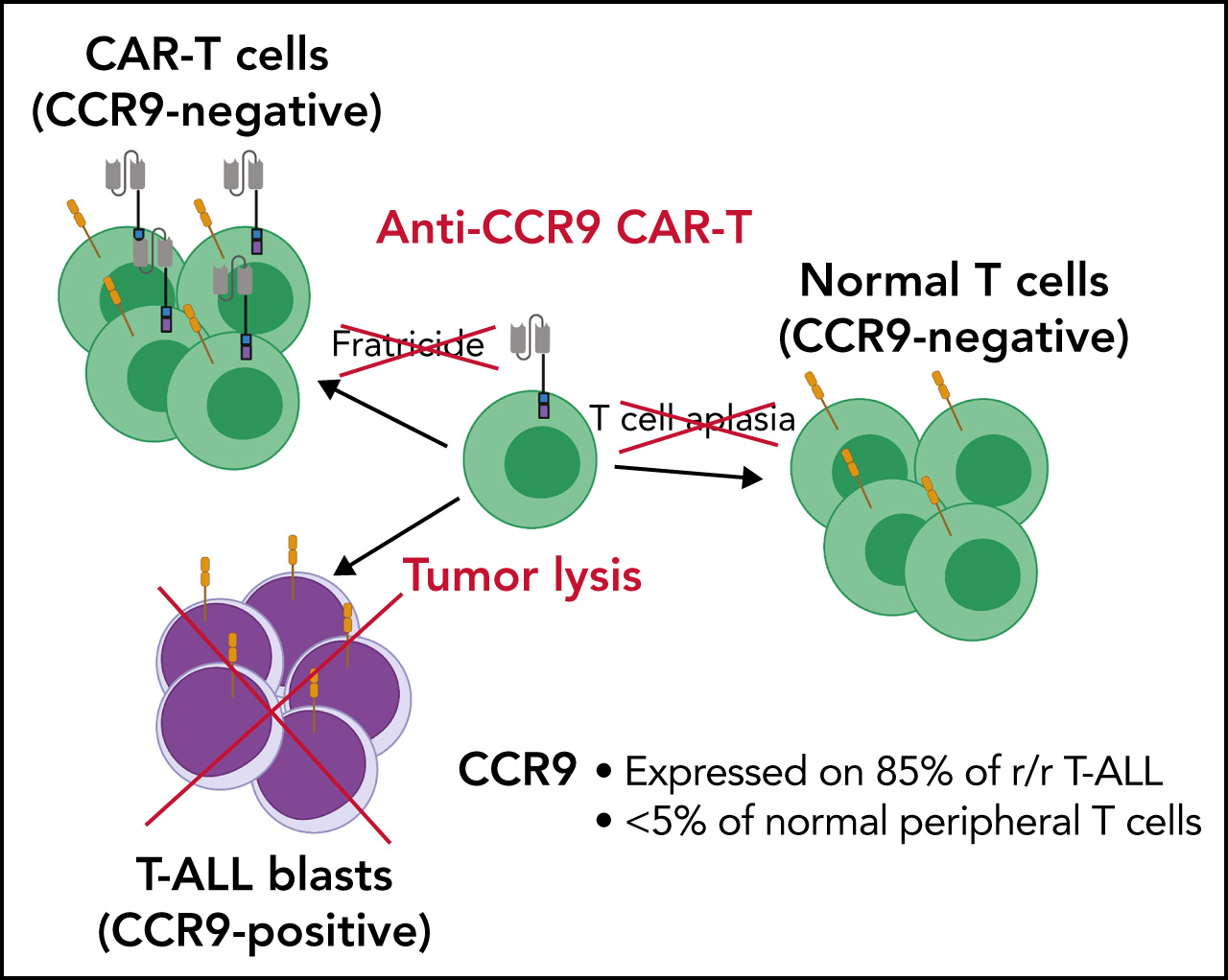
- Anti-CCR9 CAR-T cells — targeting the chemokine receptor CCR9, which is expressed on thymic-stage T-ALL but absent from most normal T cells (Blood 2022). The FRACTALL clinical trial is now testing this approach in patients.
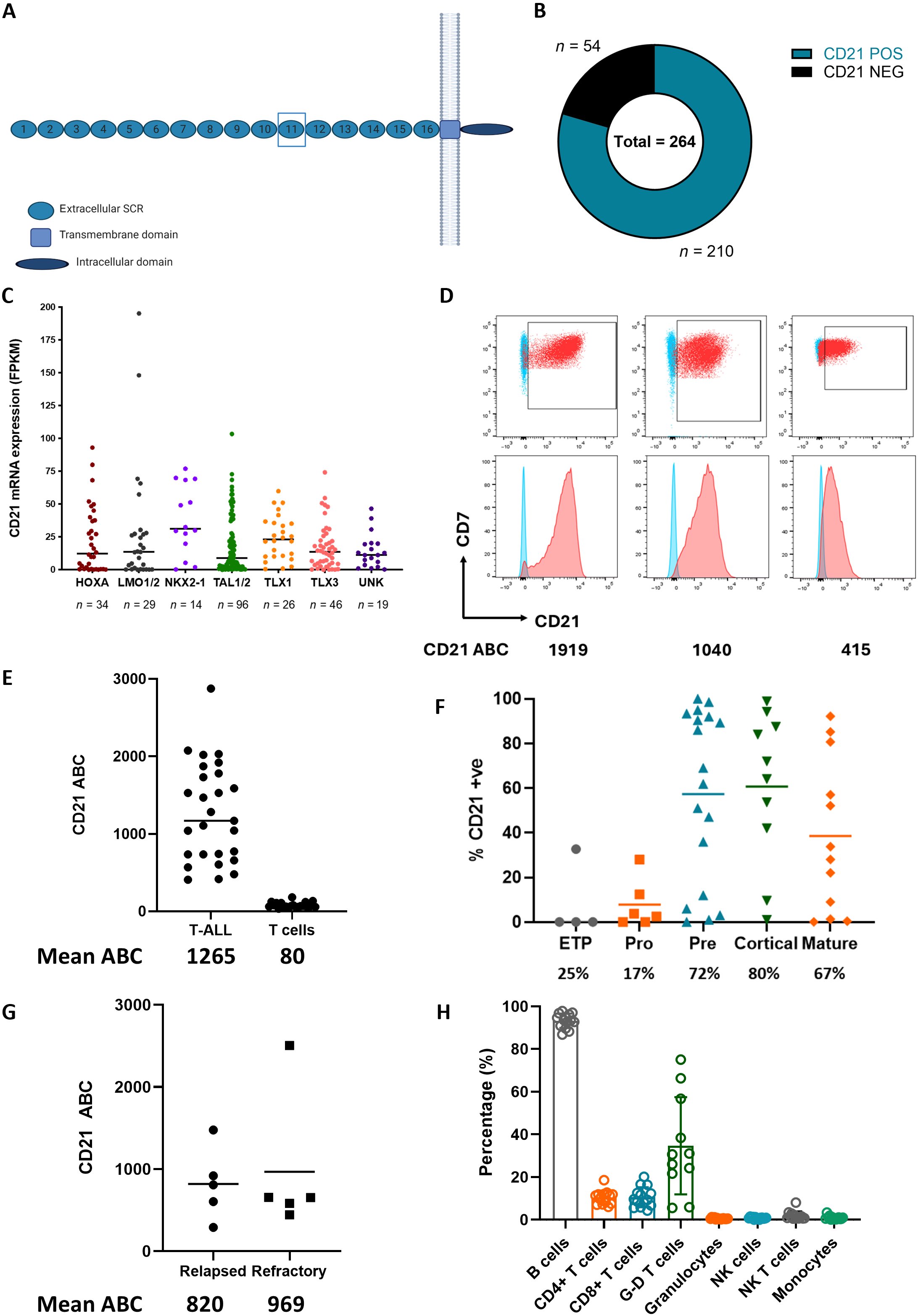
- Anti-CD21 CAR-T cells — targeting CD21, which we identified as aberrantly expressed on 80% of T-ALL cases but absent from healthy T cells (Science Translational Medicine 2025)
- Combinatorial targeting — engineering multi-antigen CARs to prevent antigen escape, a major cause of relapse after single-target therapies
- Bispecifics & ADCs — developing T cell-engaging bispecific antibodies and antibody-drug conjugates as alternative immunotherapy modalities for T-ALL
- Immunocompetent mouse models — building novel murine models of T-ALL to evaluate CAR-T efficacy in the context of an intact immune system